
by Carla Barnes | Feb 27, 2026 | Homepage Carousel, News + Events - Corporate
Portable FeNO Monitoring System Approved for a 6-Second Breath Test ~ St. Paul, MN (FEBRUARY 27, 2026) — MGC Diagnostics, a leading provider of cardiorespiratory diagnostic solutions, today announced the U.S. availability of Fenom Flo™, the only portable FeNO device...
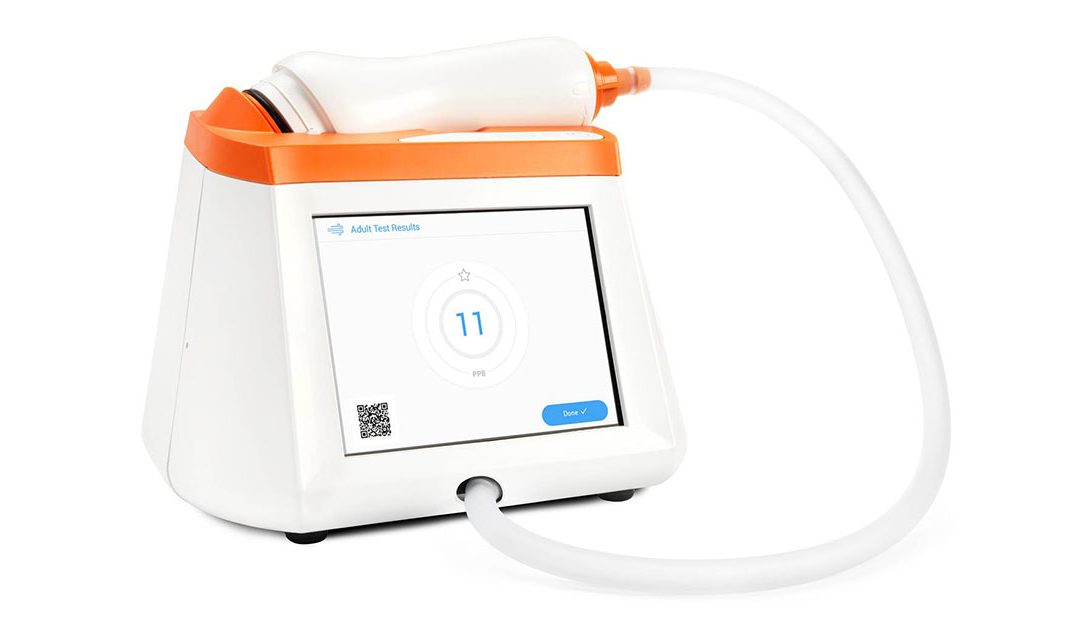
by Carla Barnes | Jun 22, 2023 | News + Events - Diagnostics, Related - Clinicians, Related - Corporate, Related - Diagnostics, Related - Patients
Major Milestone for Expanded Access to FENO Testing in US Pleasanton, CA, June 22, 2023 — CAIRE Diagnostics is pleased to announce that the Fenom Pro® can now be upgraded to include a 6-second test mode available for ages 6 and up. The completed clearance (K213611) by...

by Carla Barnes | Jun 3, 2020 | News + Events - Corporate, News + Events - Diagnostics, Related - Clinicians, Related - Commercial, Related - Corporate, Related - Diagnostics, Related - Patients, Related - Providers
Establishes CAIRE Diagnostics Inc. business unit to broaden product portfolio ~ Ball Ground, GA (June 3, 2020) – CAIRE Inc., a leader in respiratory solutions for the global health care community, announced the acquisition of Spirosure, Inc. Spirosure is a...




